Oral Mucositis
Oral mucositis is a severely debilitating condition characterized by erythema, edema, and ulcerations of the oral mucosa.
It is a complication of radiation therapy (RT) to the head and neck, chemotherapy, chemoradiotherapy, and hematopoeitic stem cell transplantation (HSCT).
Oral intake may be impaired due to pain, which in some cases may lead to the need for parenteral nutrition.
In addition, the oral lesions weaken the mucosa barrier resulting in local or systemic infection.
In severe oral mucositis caused by chemotherapy, the dose in the subsequent chemotherapy cycle may have to be decreased or delayed, affecting patients’ quality of life and worsening prognosis.
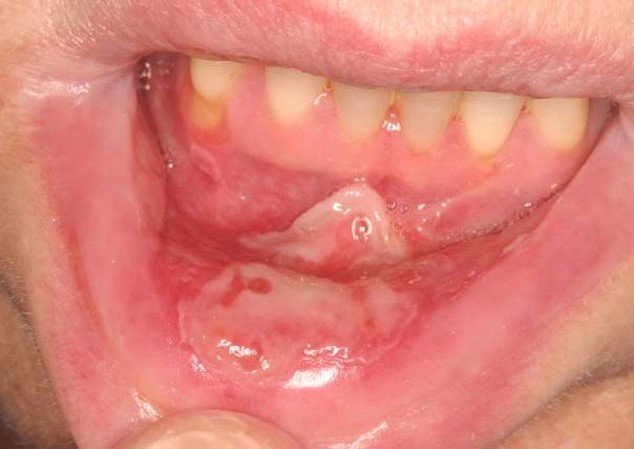
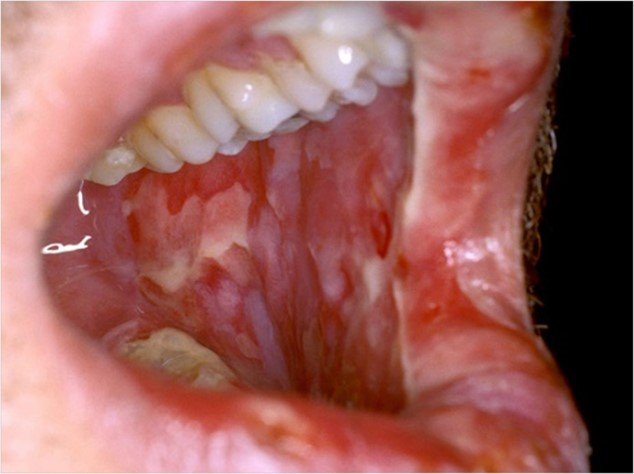
Oral mucositis is a frequent complication in patients receiving radiation therapy (RT) to the head and neck, chemotherapy for solid tumors or lymphoma, and high-dose myeloablative chemotherapy prior to hematopoietic cell transplantation.
The incidence of oral mucositis varies amongst the different chemotherapy agents.
Chemotherapeutic agents that affect DNA synthesis (S-phase), e.g., 5-fluorouracil, methotrexate, and cytarabine, have a high incidence of oral mucositis.
Anthracyclines, mTOR inhibitors, alkylating agents, and antimetabolites also have an increased risk of oral mucositis.
The basal epithelial layer of the oral mucosa has a rapid cellular turnover, making the oral epithelium susceptible to radiation injury
Cell death and the incapacity of the oral mucosa to heal lead to the development of oral mucositis.
Epidemiology - Oral Mucositis
Between 20% to 40% of patients with solid tumors receiving chemotherapy develop mucositis, usually within five to fourteen days of starting treatment.
The incidence and severity of mucositis vary between chemotherapeutic agents, the number of chemotherapy cycles, the dose of chemotherapy, and from patient to patient.
Patients who receive myeloablative preparations for hematopoeitic stem cell transplant have a higher incidence of oral mucositis.

One study reported that patients who receive high doses of chemotherapy or undergo bone marrow transplantation have a 76% risk of mucositis. Radiation-induced oral mucositis (RIOM) occurs in 100% of altered fractionation radiotherapy head and neck cancer patients.
The frequency of mucositis is higher in patients with poor nutritional status and inadequate oral care.
Younger age patients may have a higher incidence of oral mucositis.
Evaluation - Oral Mucositis
Evaluating for oral mucositis is dependent upon clinical history and physical exam findings. Laboratory and radiography are not as helpful. If ulcers are present on the hard palate, attached gingiva, or tongue dorsum, cultures should be obtained to rule out viral or fungal etiology.
The severity of mucositis is measured on a well-defined scale, and several different scales have been developed
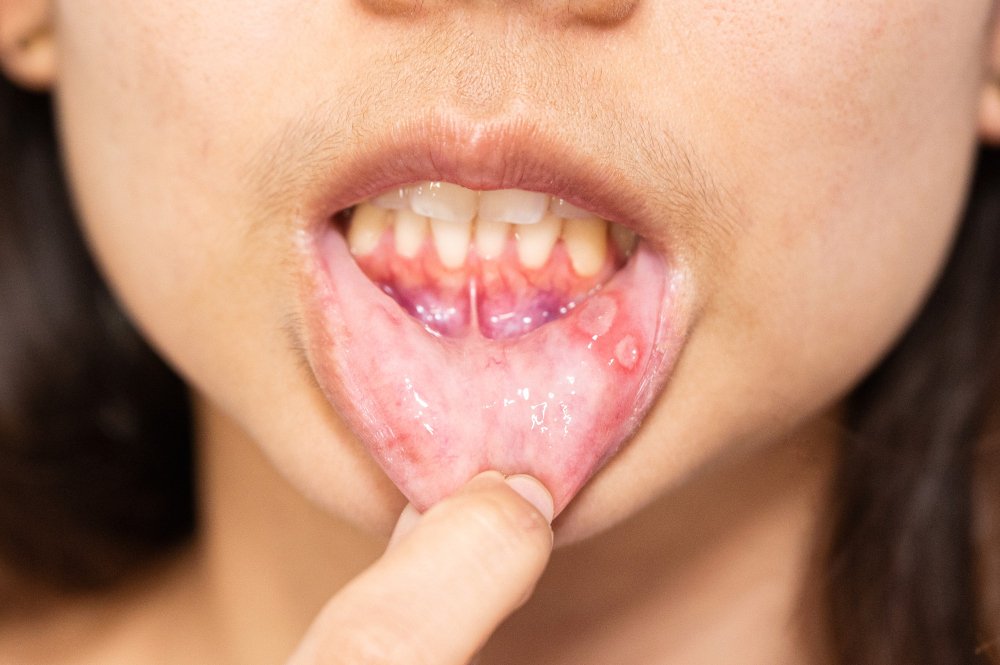
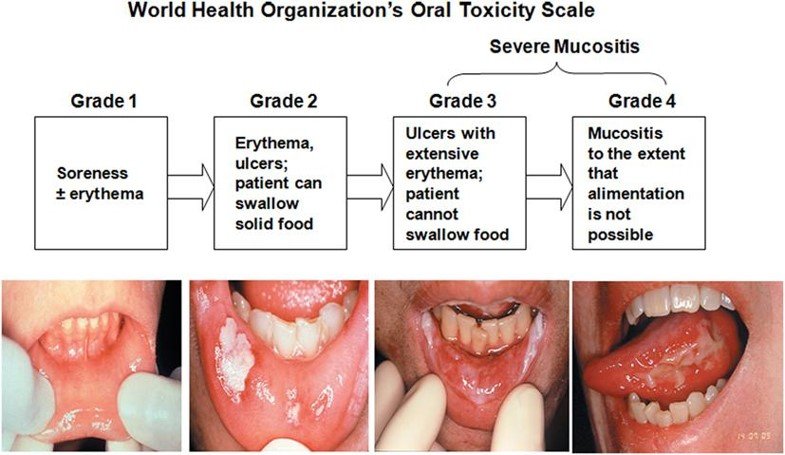
World Health Organization (WHO)
The World Health Organization (WHO) scale combines both subjective and objective measures of oral mucositis.
- Grade 0 = No oral mucositis
- Grade 1 = Erythema and soreness
- Grade 2 = Ulcers, able to eat solids
- Grade 3 = Ulcers, requires a liquid diet (due to mucositis)
- Grade 4 = Ulcers, alimentation not possible (due to mucositis)
Oral Mucositis – Limitations of Current Therapies
Pain is a serious problem with oral mucositis, especially in the mouth. Topical agents may not last long in the mouth, or may not be able to reach all the affected areas.
Pain management requires a combination of different approaches to manage pain, including topical gels and mouthwashes, over-the-counter pain medications and even prescription opioids.

Pain treatments for mucositis include:
Topical anesthetics
Some of these include lidocaine, benzocaine, dyclomine hydrochloride (HCl) etc
Magic mouthwash
A mix of lidocaine and diphenhydramine , Anti-inflammatory drugs, such as prednisone.
Acetaminophen (paracetamol)
Prescription narcotic pain medications
Almost all of the currently available options for Oral Mucositis barely manage the pain, if at all and no effect whatsoever on healing and recovery
MUCOFAN™ Composition

Long Term I.P. Protection for Mucofan – U.S. Patent under application
Significantly cost effective and highly effective for Oral Mucositis
API has GRAS notification from US FDA
API (Lactobacillus brevis CD2) is Approved as Orphan Drug in Bechet’s Disease by US FDA
